Chlorofluorocarbons (CFCs) and hydrochlorofluorocarbons (HCFCs) have historically been used in a variety of refrigeration applications in manufacturing, from large process chillers to small office air conditioning units. Sections 608 of the Clean Air Act and subsequent federal regulations under 40 CFR 82 regulate these refrigerants to minimize detrimental atmospheric changes caused by releases. As recent regulations have changed, it is important to understand the mechanisms by which the refrigerants can cause atmospheric changes and how to manage facility use.
Understanding Ozone
Ozone is a highly reactive compound comprised of three oxygen molecules, represented by the chemical formula O3. Most atmospheric ozone is concentrated as a layer in the stratosphere and is formed naturally through a photochemical reaction between molecular oxygen and solar ultraviolet radiation. Stratospheric ozone is essential to protecting life on earth as it absorbs biologically harmful ultraviolet B (UV-B) radiation, preventing it from reaching the Earth’s surface in harmful quantities. Stratospheric ozone is different from tropospheric, or ground-level ozone, which is primarily formed through photochemical reactions of nitrogen oxides and volatile organic compounds emitted from man-made sources (fossil fuel combustion, industrial activities). Tropospheric ozone is a primary constituent of photochemical smog. Stratospheric ozone is often referred to as “good” ozone due to its important role in absorbing UV-B radiation. Tropospheric ozone is often referred to as “bad” ozone as it causes respiratory issues in animals and negatively affects plant photosynthesis and respiration.
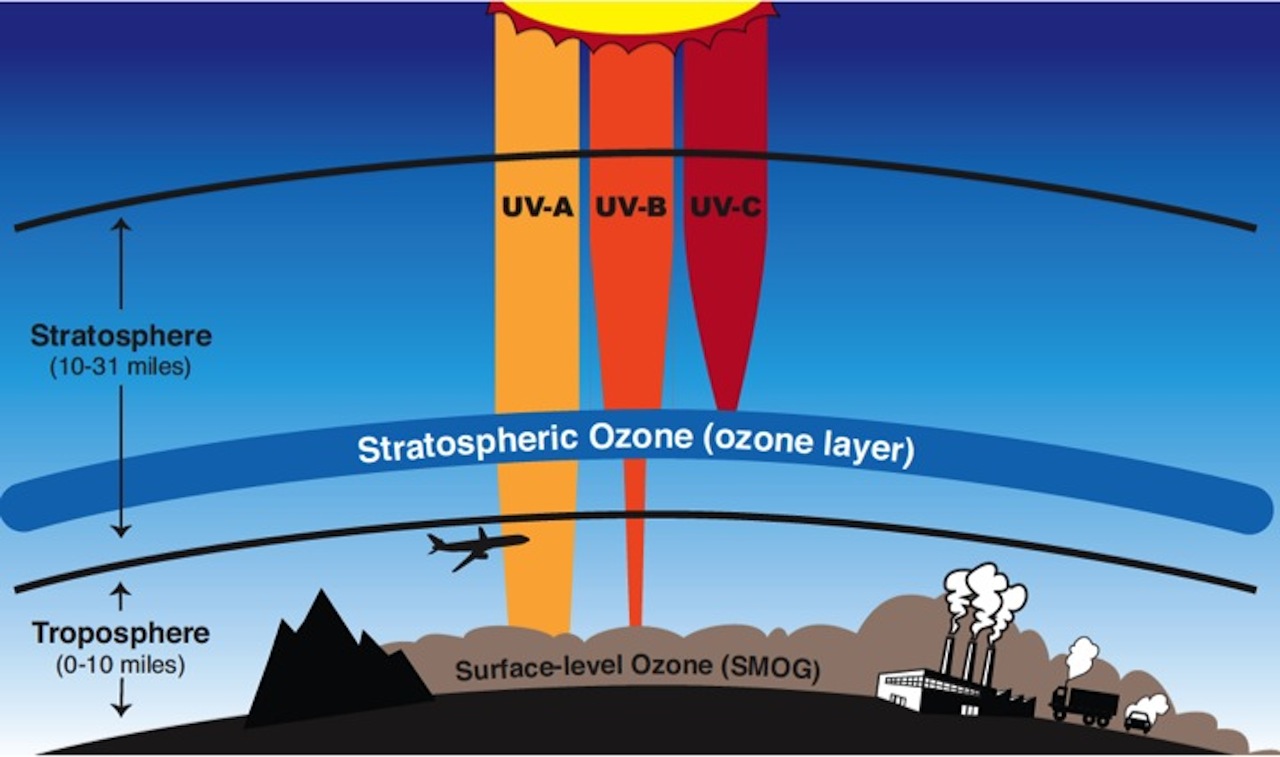
Figure 1: Stratospheric ozone vs. tropospheric ozone. Source: NASA
Mechanism of Ozone Depletion
The primary cause of stratospheric ozone depletion results from the introduction of manufactured halocarbons into the Earth’s upper atmosphere. Sources of halocarbon emissions at the Earth’s surface include refrigeration leaks, solvent use, the manufacturing of certain foam products, and propellants. Once emitted, the halocarbon molecules are transported to the stratosphere through turbulent mixing in the troposphere, the atmospheric layer that extends from the Earth’s surface to the stratosphere. The halocarbon molecules undergo a photochemical reaction in the stratosphere that results in the release of halogen ions, which subsequently act as catalysts to the dissociation of ozone molecules into oxygen molecules:
CFCl3 + ultraviolet light → Cl + CFCl2
Cl + O3 → ClO + O2
ClO + O3 → Cl + 2O2
These reactions can reoccur over 100,000 times with a single halogen ion before the ion is removed from the stratosphere. Halocarbons, particularly chlorine, accelerate the depletion of stratospheric ozone at a faster rate than it can naturally regenerate. Hydrofluorocarbons (HFCs) also contribute to ozone depletion, but at a much smaller scale than CFCs and HCFCs. The primary cause of atmospheric changes by HFC emissions is through the absorption of heat in the stratosphere by HFC molecules, which increases stratospheric and tropospheric temperatures. CFCs, HCFCs, and HFCs are all heat-trapping gases, with global warming potentials 100 to 10,000 times greater than carbon dioxide (CO2).
Refrigeration Management Regulations
In 1990, Title VI of the Clean Air Act was enacted by the U.S. Congress to add provisions for protecting stratospheric ozone. This provided a regulatory basis to implement the Montreal Protocol. Title VI, Section 608, and the resulting federal regulations set timelines for the phaseout of CFCs, HCFCs, and other ozone-depleting substances (ODS). It also established requirements for proper management of ODS to prevent or mitigate releases. A November 2016 amendment to Section 608 and 40 CFR 82 enacted the regulation of HFCs in the same manner as ODS, and reduced leak rate thresholds that trigger repair, retrofit, or replacement requirements. Industry groups challenged the EPA’s authority to extend regulations on ODS to non-ozone-depleting substitute refrigerants with high global warming potential (GWP). As a result, the November 2016 amendment was rescinded and replaced in February 2020. The current federal rule requirements are summarized below.
For appliances with 50 or more pounds of ODS per individual circuit:
- Maintain annualized leak rate tracking;
- Repair or replace appliances that leak above the following leak rate thresholds:
- 35% for Industrial Process and Commercial Refrigeration Units,
- 15% for Comfort Cooling Equipment;
- Conduct verification tests on repairs;
- Periodically inspect for leaks;
- Report chronically leaking appliances to the EPA;
- Retrofit or retire appliances that are not repaired; and
- Maintain related records.
For appliances with any amount of ODS or non-ODS high GWP replacement refrigerants (new with the 2020 rule):
- Purchasers or handlers must be EPA/Section 608 certified;
- Anyone removing refrigerant from a refrigeration appliance must evacuate refrigerant to a set level using certified refrigerant recovery equipment before servicing or disposing of the appliance;
- The final disposer (such as scrap recyclers or landfills) must ensure and document that refrigerant is recovered; and
- All used refrigerant must be reclaimed to industry purity standards before it can be sold to another appliance owner.
Sources
Vallero, D. Fundamentals of Air Pollution, 5th ed. 2014. pp. 69-72. Academic Press, Waltham, MA. Schlesinger, W.H. Biogeochemistry – An Analysis of Global Change, 2nd edition 1997. Academic Press, San Diego, CA. United States Environmental Protection Agency, Basic Ozone Layer Science. https://www.epa.gov/ozone-layer-protection/basic-ozone-layer-science United States Environmental Protection Agency, Understanding Global Warming Potentials https://www.epa.gov/ghgemissions/understanding-global-warming-potentials


